|
Fill a graduated cylinder with a known volume of water, then carefully immerse the object in the cylinder. Archimedes’ Principle: For irregularly shaped objects, you can use Archimedes’ principle to measure the volume indirectly.Follow the instructions for proper usage and record the volume accordingly. These instruments are especially useful in laboratory settings where accuracy is crucial. Pipette or Burette: A pipette or burette can be used to measure precise volumes of water.Pour the water into the cylinder, making sure to read the measurement at eye level, where the bottom of the meniscus aligns with the graduation marks. Graduated Cylinder: Use a graduated cylinder to measure the volume of water.There are several methods to measure the volume of water, depending on the equipment available and the precision required. Place a container on the scale and tare it to zero, then carefully pour the water into the container. Ensure that the balance or scale is calibrated properly before taking any measurements. To measure the mass of water, you can use a balance or a scale. How to Find Density of Water Measuring Mass To calculate the density of water, we need to determine the mass and volume of the water sample. The density of water, in particular, refers to the mass of water contained in a specific volume. In simpler terms, it measures how tightly packed the particles are in a substance. In order to perfectly understand how to calculate the density of water, we need to understand the definition of density and its formula below: What is Density?ĭensity is a fundamental property of matter that represents the amount of mass within a given volume. Therefore, the density of water is 1 gram per millilitre Definition of Density, its Formula, and SI Unit How to Calculate Density of Water Exampleįor example, if the mass is 9 g and the volume is 9 mL. Ensure to add the si unit to the final result which is in g/mL or gcm -3. Third Step (Solution): Now, you substitute your formula for the density of water with the data available to you and solve the problem. 
Second Step (Formula): In this stage, you state the equation that will help you to calculate the density of water, which is Density (ρ) = Mass (m) / Volume (v) or ρ=m/v. Here are three steps that will help us in calculating the density of water:įirst Step (Data): It is important to read and understand the question so as to gather all the necessary information. How to Calculate Density of Water: Step-by-Step Guide Ensure consistent units, such as grams for mass and cubic centimetres for volume, or kilograms for mass and cubic meters for volume, to obtain the density in the appropriate units. Then, plug these values into the formula and perform the division to find the density. Measure the mass of the water sample using a scale and the volume using a graduated cylinder or similar tool. To calculate the density of water, you use the formula: The density of water is approximately 1 gram per cubic centimetre (g/cm³) or 1000 kilograms per cubic meter (kg/m³) at standard atmospheric pressure and temperature. Conclusion What is the Density of Water and How to Calculate it?.Why is knowing the density of water important? Is the density of water the same everywhere on Earth? 
How does salinity affect the density of water? 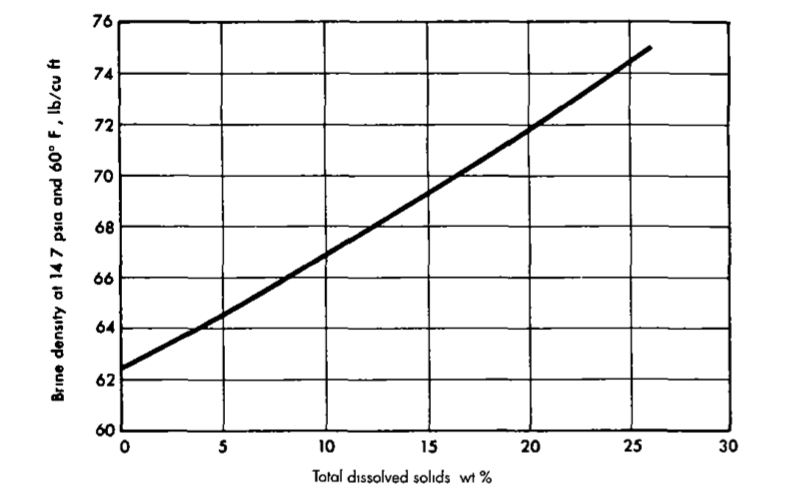
Can the density of water be greater than 1? How does the density of water change with temperature? FAQs about Calculating the Density of Water.Formula for Calculating Density of Water.Definition of Density, its Formula, and SI Unit.How to Calculate Density of Water: Step-by-Step Guide. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
